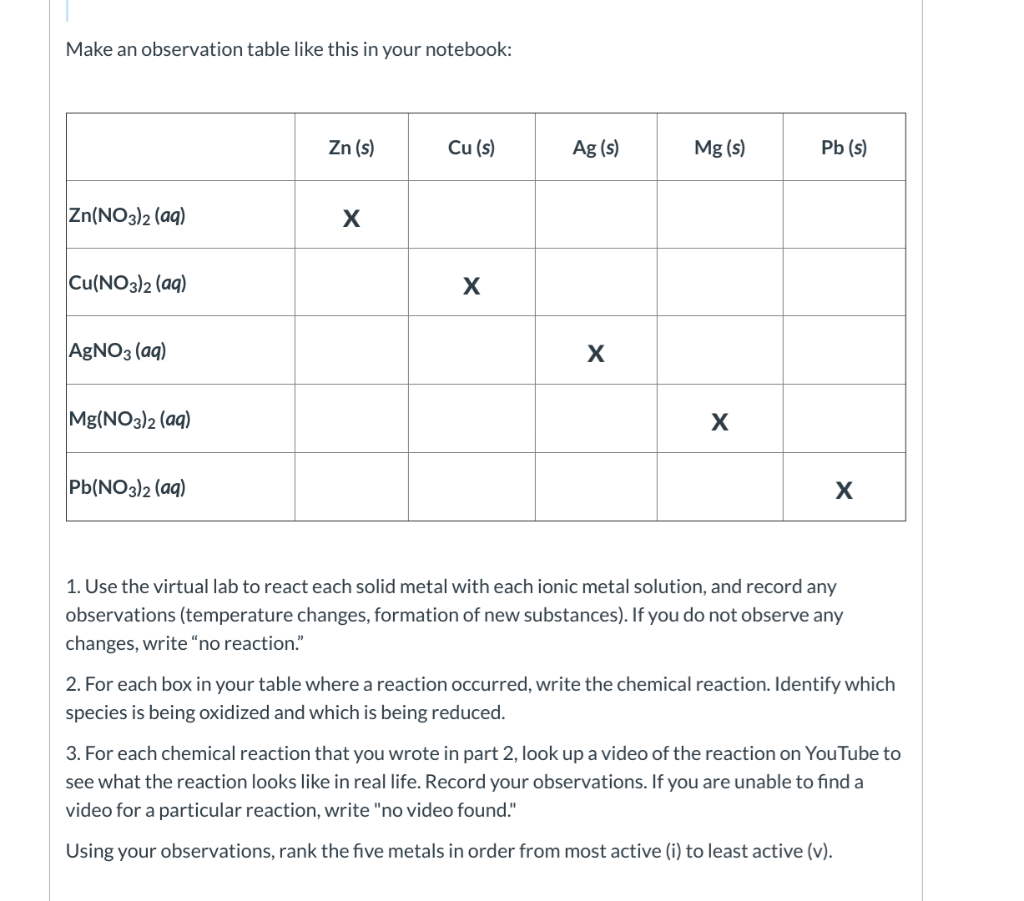
Students should also explain any discrepancies between their results and the publish information. from both activities, into a single Activity Series of Metals. Also, the published version of the activity series may be compared to the results. Click on Activity 2, and follow the same procedures with the new metals and solutions. Once this results have been compiled as a class, discuss how the activity aligns with the metals They should be able to justify their decision. Metals are usually reclaimed from their ores in one of two ways: chemical reduction (using carbon in the form of coke) or electrolytic reduction (using electricity). The students will be instructed that they are to determine the activity of each of the metals and rank them according to their results. The materials needed for this lab include 4 metal samples- such as magnesium, iron, copper, zinc, and tin-spot plates/test tubes, 0.1M HCl or 1.0M HCl, and a container for disposal of waste products. To determine the reactivity series of common metals to observe single replacement reactions. This will lead to the objective of this lab to determine the activity of four mystery metals. Pre-lab set up can occur as recently as the day before even though the lab. Lab-Activity Series Of Metals Activity of 3 metals observed by students who then must classify them in order of most active to least active. 
An Activity Series is provided at the end of the Background section. An Activity Series of elements is often used to determine if (A) will displace (B) in a single displacement reaction. If it is a gas, it will appear as bubbles. The metal wires from Part B should be returned to the set-up sheet to be used by the next lab section.' The metals from Part A2 of the experiment should be removed from the reactions with forceps, rinsed with water if they have been exposed to acid, blotted to remove excess water, then discarded in the container for used metals. We will develop the activity series by testing different metals reactivity by. If the new element (B)is a metal, it will appear as a metallic deposit. As students discuss that reasons for the production of hydrogen, ask whether any metal combined with HCl will produce this reaction. Activity Of Metals Lab A microscale activity of metals lab where students develop an activity series based on their observations. The purpose of this lab was to develop an activity series of the elements. EXPERIMENT 8 Activity Series (Single Displacement Reactions). A possible anticipatory set for this activity would be the demonstration of the hydrogen "pop" test. NOTE-YOU MAY OMIT PUTTING METAL INTO ITS OWN SALT SOLUTION, SEE DATA SEET 3.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
